Philip J. Dutton
Organic Chemistry
Ph.D. (Victoria)
dutton@uwindsor.ca
Associate Professor
Department Head
253-3000 Ext. 3526
273-2 Essex Hall
Personal Webpage
RESEARCH INTERESTS:
Supramolecular chemistry is the chemistry of the non-covalent bond. Non-covalent interactions include most molecular associations and the forces that may be involved can range from very weak London forces to strong interactions such as hydrogen bonds and ion dipole interactions.
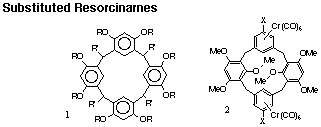
We carry out studies in the latter two areas. Ion-dipole interactions have been explored using the calix[4]resorcinarene system as shown in the figure. The octa-substituted species was synthesized in this laboratory and a wide variety of physical characterization has been carried out. Work in this area has quantified the association constants of the resorcinarnes toward a variety of metal cations, characterized the Langmuir-Blodgett (LB) film behaviour of these species and identified energy transfer luminescence processes in solutions and LB films. Work is ongoing to prepare new resorcinarene materials that incorporate a second binding site in the molecule in order to exploit the energy transfer luminescence characteristics in the development of chemical sensor devices.
Our second avenue of approach to synthetic host molecules is directed toward hydrogen bonding host molecules. After a series of difficulties, we have succeeded in synthesizing six precursors to hydrogen bonding host systems, two of which are shown below.
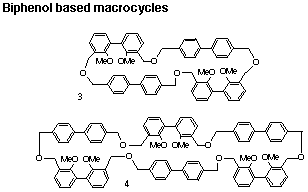
Now that the major cyclization hurdle has been crossed, the refinement to build operating host molecules is underway. These types of compounds incorporate two convergent hydrogen bonding sites held apart by rigid spacers. There is a vast amount of work to do in this exciting area and Ph.D. students will find this a challenging and rewarding field of effort.

